Abstract
Proteolysis Targeting Chimeras (PROTACs) are bifunctional molecules that recruit an E3 ubiquitin ligase to a protein of interest (POI), catalyzing its ubiquitination and proteasomal degradation. Because they remove target proteins rather than simply inhibit them, PROTACs offer potential advantages for targeting “undruggable” proteins, overcoming drug resistance, and improving selectivity. This article reviews the mechanism of PROTACs, design considerations, recent advances including undergoing clinical trials, and current challenges.
- Introduction
Traditional small-molecule drugs function primarily by inhibiting protein activity, yet nearly 80% of disease-related proteins lack suitable binding pockets [1]. This leaves many proteins “undruggable” using classical approaches.
PROTACs provide a paradigm shift: rather than inhibiting proteins, they eliminate them by hijacking the ubiquitin–proteasome system (UPS). The first PROTAC, reported in 2001, targeted MetAP-2 using a peptide ligand linked to an SCF E3 ligase ligand [1]. Since then, hundreds of PROTACs have been designed against kinases, receptors, transcription factors, and aggregation-prone proteins.
- Mechanism of Action
A PROTAC molecule has three parts:
- POI ligand (warhead) – binds the target protein.
- E3 ligase recruiter ligand – binds to an E3 ligase (commonly cereblon (CRBN) or Von Hippel–Lindau (VHL)) [2].
- Linker – a flexible chain that connects the two ligands.
Here’s the annotated PROTAC structure with
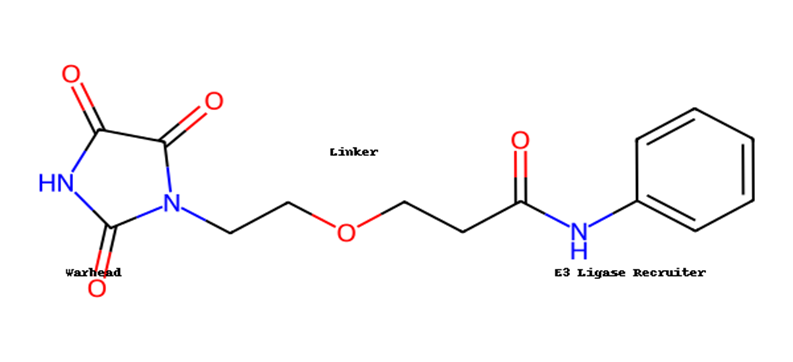
the three key domains labeled
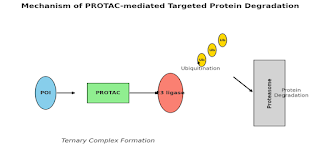
Mechanism:
- The PROTAC simultaneously binds the POI and E3 ligase, forming a ternary complex.
- The E3 ligase transfers ubiquitin chains to the POI.
- The ubiquitinated protein is degraded by the 26S proteasome.
- The PROTAC is released and can catalyze further degradation rounds [1,2].
- Advantages of PROTACs
- Degrade rather than inhibit: Removes both catalytic and scaffolding functions of the protein [3].
- Catalytic mode of action: A single PROTAC can degrade multiple POI molecules [3].
- Expanded target space: Enables targeting of transcription factors, scaffolding proteins, and receptors without active sites [4].
- Overcoming resistance: Effective against mutant proteins that resist conventional inhibitors [2].
- Improved selectivity: By exploiting ternary complex formation, PROTACs may show higher selectivity than inhibitors [5].
4. Clinical Progress
Several PROTACs have advanced into clinical trials:
| Compound | Target | Indication | Phase | Highlights |
| ARV-471 (Vepdegestrant) | Estrogen receptor (ER) | ER+/HER2– breast cancer | III | Showed superior PFS in ESR1-mutant subgroup [6]. |
| ARV-110 (Bavdegalutamide) | Androgen receptor (AR) | Prostate cancer (mCRPC) | I/II | Demonstrated activity in AR-mutant tumors [2]. |
| ARV-766 (Luxdegalutamide) | AR | Prostate cancer | I/II | Strong activity in resistant disease [2]. |
| BMS-986365 (CC-94676) | AR | Prostate cancer | III | Early trials show PSA reduction [6]. |
Over 25 PROTACs are currently in human trials, most based on CRBN recruitment [2].
5. Challenges
- Drug-like properties: Large molecular weight, poor solubility, and limited permeability hinder oral bioavailability [7].
- Off-target degradation: Promiscuous degradation due to unintended interactions [2].
- E3 ligase limitations: Current reliance on a small set of ligases (CRBN, VHL); tissue-specific ligases remain underutilized [1,4].
- Synthetic complexity: Optimization of warhead, linker, and ligase ligand is resource-intensive [5].
6. Future Perspectives
- Expanding E3 ligase toolbox: Identification of new ligases with tissue-specific expression [4].
- Next-generation degraders: Including LYTACs (lysosome-targeting chimeras) for extracellular proteins [1].
- Computational design & AI: Predicting ternary complex stability to accelerate PROTAC discovery [7].
- Beyond oncology: Applications in neurodegeneration, immunology, and viral infections [3].
7. Conclusion
PROTACs mark a revolution in therapeutic strategy—from static inhibition to dynamic protein elimination. With multiple agents in late-stage clinical trials, they are poised to become integral tools in precision medicine. Continued improvements in design, delivery, and safety will determine their success as mainstream therapies.
References
[1] Zhao L., Zhao J., Zhong K., Tong A., Da J. Targeted protein degradation: mechanisms, strategies and application. Signal Transduction and Targeted Therapy. 2022;7:113. doi:10.1038/s41392-022-00966-4.
[2] Zhong G., et al. Targeted protein degradation: advances in drug discovery. Signal Transduction and Targeted Therapy. 2024. doi:10.1038/s41392-024-02004-x.
[3] Békés M., et al. PROTAC targeted protein degraders: the past is prologue. Nat Rev Drug Discov. 2022;21:181-200. doi:10.1038/s41573-021-00371-6.
[4] Ocaña A., Pandiella A. Proteolysis targeting chimeras (PROTACs) in cancer therapy. J Exp Clin Cancer Res. 2020;39:189. doi:10.1186/s13046-020-01672-1.
[5] Vicente A.T.S., et al. Synthesis, biological evaluation and clinical trials of cereblon-based PROTACs. Nat Commun. 2025. doi:10.1038/s42004-025-01598-9.
[6] BiochemPEG. The latest progress of PROTAC degraders in clinical trials. 2024. Available at: https://www.biochempeg.com/article/434.html
[7] Liu Z., et al. An overview of PROTACs: a promising drug discovery paradigm. Front Chem. 2022;10:1032394. doi:10.3389/fchem.2022.1032394.
